When tasked with the job of building a cage, there are a few things that first come to mind. Cube, six sides, material, bars… captivity? But it can’t be that bad, right? When tasked with building a pollinator exclusion cage… how different can it be? We discovered that building a cage, any type of cage, is a lot harder than it sounds, at least when one is building their very first cage.
First comes the task of design; we drew out the sides of the cage. Four sides, six sides, five sides? Soon there were fifteen rectangles on a paper, arrows pointing from one to the other, dark lines and light lines indicating different “theoretical” material. Redesign, was inherent. And so, there was over complication, and more over complication, and then simplification and more simplification, before we found what we were looking for, the basics.
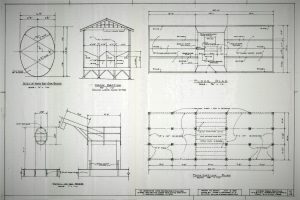
(Images taken from Google)
Acquiring materials turned out to be a long process with Lowe’s not being the most familiar of places. Finding wood, making a decision about dimensions, getting said wood cut, searching for flat L’s, locks, hooks, glue, tulle, staples, a staple gun (which initially did not work, and so a replacement was required… like all good things), and finally the motivation to put all the little pieces together. Me and Jack worked Monday and Tuesday, with a bit of anticipation, as we attempted to create our first pollinator exclusion cage. I can say, I got a little carried away with the staple gun when initially applying tulle, but Jack was able to remedy the 100-staple frames to a lot less.
Finally, after much work and some improvised super glue patchwork (tulle rips surprisingly easily, who knew?) we were a able to complete the first pollinator exclusion cage. It truly was a masterpiece. And then we had to build the second one. Jack and Ashley traveled to Lowe’s today to resupply, and then we set off on the second session of construction. Ashley and Elizabeth helped me and Jack apply to tulle to the frames, on one of the windiest days of the week (perfect timing), making our lives much, much easier. And so we worked and completed the second pollinator exclusion cage, this one with a minimal amount of super glue (E-6000).


So we learned, that the building of cages is a task of trial and error. And that as always, practice makes perfect. It was truly a learning experience. Now, we endeavor to use these cages in the next task of keeping pollinators away from C. americana (Bellflower), while at the same time exposing our plant of study to the surrounding environment.